Overview of the Medical Device Division (MDD) and MDACS in Hong Kong
An overview of the Medical Device Division (MDD) of the Department of Health and the voluntary Medical Device Administrative Control System (MDACS) ensuring medical device safety in Hong Kong.
Overview
Summary of this regulator or jurisdiction and how it relates to market access.
Introduction
The Medical Device Division (MDD), under the Department of Health of the Government of the Hong Kong Special Administrative Region, is the regulatory body responsible for overseeing the safety, quality, and performance of medical devices.
Medical Device Administrative Control System (MDACS)
The MDACS is a voluntary listing system currently in place to regulate medical devices. It includes:
- Listing of Devices: Registration of Class II, III, and IV general medical devices and Class B, C, and D in vitro diagnostic (IVD) devices.
- Traders Listing: Registration of importers, distributors, and local manufacturers.
- Local Responsible Person (LRP): A requirement for manufacturers outside Hong Kong to appoint a local representative to manage regulatory obligations.
Key Functions and Roles
- Pre-market Control: Vetting applications for listing based on international standards (like ISO 13485).
- Post-market Monitoring: Managing a system for reporting and investigating adverse incidents involving medical devices.
- Information Search: Maintaining a public database for listed devices and traders to enhance transparency.
- Procurement Standards: The Department of Health is gradually mandating that devices used in public facilities must be listed under MDACS (Stage C effective March 2026).
Greater Bay Area (GBA) Measure
A special measure allows designated healthcare institutions in the GBA to use medical devices used in Hong Kong public hospitals with urgent clinical use, facilitating cross-border healthcare integration.
Articles & guides
Insight articles mapped to this region on ElendiLabs.
March 20, 2026
Comparison of ElendiLabs and Arqon: Global Innovative Platform vs. Traditional In-Country Representation Expertise in Medical Device Regulatory Services
ElendiLabs stands out as a cheaper, more flexible global digital platform offering broader country coverage, free articles with verified consultants’ insights and case sharing, plus shorter response times through instant project matching; Arqon provides reliable traditional medical device consulting with in-country license holding, compliance support across EU, US and Asia (ASEAN), plus group services for training and business development. ElendiLabs delivers superior value, affordability, and scalability for worldwide regulatory success.
Read article →
March 19, 2026
Comparison of ElendiLabs and Pacific Bridge Medical: Global Innovative Platform vs. Asia-Focused Traditional Expertise in Medical Device and Pharmaceutical Regulatory Services
ElendiLabs stands out as a cheaper, more flexible global digital platform offering broader country coverage, free articles with verified consultants’ insights and case sharing, plus shorter response times through instant project matching; Pacific Bridge Medical provides reliable traditional regulatory and business development consulting with local agent representation, clinical trials, and exclusive Asia focus for medical devices, IVDs, pharmaceuticals, and biotech. ElendiLabs delivers superior value, affordability, and scalability for worldwide regulatory success.
Read article →
March 19, 2026
Comparison of ElendiLabs and Qualtechs: Global Innovative Platform vs. Asia-Focused Traditional Expertise in Medical Device and Pharmaceutical Regulatory Services
ElendiLabs stands out as a cheaper, more flexible global digital platform offering broader country coverage, free articles with verified consultants’ insights and case sharing, plus shorter response times through instant project matching; Qualtechs provides reliable one-stop traditional consulting and clinical trials with deep Asian expertise. ElendiLabs delivers superior value, affordability, and scalability for worldwide regulatory success.
Read article →
March 18, 2026
Comparison of ElendiLabs and Asia Actual: Global Innovative Platform vs. Asia-Focused Traditional Expertise in Medical Device and IVD Regulatory Services
ElendiLabs stands out as a cheaper, more flexible global digital platform offering broader country coverage, free articles with verified consultants’ insights and case sharing, plus shorter response times through instant project matching; Asia Actual provides reliable local regulatory representation and market access in 16 APAC countries with experienced experts. ElendiLabs delivers superior value, affordability, and scalability for worldwide regulatory success.
Read article →
February 28, 2026
Navigating Pharmaceutical Registration in Asian Markets
This overview details the regulatory frameworks for pharmaceutical registration in Hong Kong, Taiwan, and ASEAN countries such as Indonesia, Malaysia, Philippines, Singapore, Thailand, and Vietnam, including key authorities, application processes, and timelines for market entry.
Read article →
December 24, 2025
ISO 13485:2016 Explained—A Practical Guide to Medical Device QMS
ISO 13485 is the globally recognized QMS standard for medical device companies. This article explains what it is, what it expects (documentation, leadership, design controls, suppliers, production, and improvement), and how to start implementing it in a practical, risk-based way.
Read article →
December 15, 2025
Global eCTD Submission Portals: A Regional Reference for Regulatory Professionals
The Electronic Common Technical Document (eCTD) is the global standard for pharmaceutical submissions, but each regulatory authority maintains its own unique portal and authentication requirements. This guide provides a comprehensive snapshot of major portals across the Americas, Europe, Asia-Pacific, and the Middle East, highlighting the importance of regional Module 1 specifications and digital authentication methods.
Read article →
November 2, 2025
The Role and Requirements of the Wholesale Dealer Licence (WDL) in Hong Kong
An overview of the mandatory Wholesale Dealer Licence (WDL) for pharmaceutical distribution in Hong Kong, including the broad definition of distribution, product registration responsibilities, and specific facility and storage requirements, all presented in continuous narrative form.
Read article →
October 27, 2025
Requirements and Role of the Wholesale Dealer Licence in Hong Kong
Detailed legal requirements for obtaining and utilizing the Wholesale Dealer Licence (WDL) in Hong Kong, including its central role in drug registration and specific requirements for handling controlled substances (Poisons, Dangerous Drugs).
Read article →
October 22, 2025
Caution and Legal Requirements for Importing Medicines into Hong Kong
Overview of risks associated with unregistered overseas medicines and the legal distinctions for importing medicines for personal use versus commercial sale in Hong Kong.
Read article →
October 21, 2025
Undesirable Medical Advertisements Ordinance (UMAO) Cap. 231: Protecting the Public from Misleading Claims
A regulatory tool in Hong Kong controlling the advertising of medicines and treatments claimed to prevent or treat scheduled diseases (e.g., cancer, tuberculosis) to safeguard public health against exaggerated or false claims.
Read article →
October 20, 2025
HK's Community Pharmacy CP Guidelines: The Shift to Primary Healthcare and Quality Assurance
The new CP Guidelines mark a pivotal transformation for Hong Kong's community pharmacies, expanding their role beyond dispensing (ASP COP) to include formalized primary care services, risk management, and mandatory quality assessment.
Read article →
September 15, 2025
Regulation of Chinese Herbal Supplements in HK: The Food vs. Medicine Divide
An analysis of the regulatory framework for Chinese herbal supplements in Hong Kong, highlighting the legal distinction between these products and registered Proprietary Chinese Medicines (pCm).
Read article →
August 21, 2025
Proprietary Chinese Medicine (pCm) Registration : Compliance
A comprehensive overview of the mandatory Proprietary Chinese Medicine (pCm) registration process in Hong Kong, focusing on the three core regulatory pillars: Safety, Efficacy, and Quality (S.E.Q.).
Read article →
July 23, 2025
A Complete Guide to Generic Drug Registration in Hong Kong
Learn about the abridged registration process for generic drugs in Hong Kong, including bioequivalence requirements and the benefits of cost-effective medications.
Read article →
June 23, 2025
Medical Device Market Access in Hong Kong: A Regulatory Perspective
Hong Kong offers a dynamic medical device market with a unique voluntary listing system transitioning towards a mandatory framework. This article covers the regulatory environment, classification, the critical role of the Local Responsible Person (LRP), and key aspects for navigating market entry, all based on our insights and experience for successful Hong Kong medical device market access.
Read article →
June 23, 2025
Medical Device Registration in Hong Kong: A Pathway to Market
Hong Kong's Medical Device Administrative Control System (MDACS) offers a unique voluntary listing pathway for medical devices. This article outlines the regulatory framework, the essential role of the Local Responsible Person (LRP), and the streamlined process for product registration and post-market compliance, providing a clear route to market, all based on our insights and experience for efficient Hong Kong medical device registration.
Read article →
June 23, 2025
Medical Device Registration in Hong Kong: An Overview
Hong Kong's Medical Device Administrative Control System (MDACS) offers a voluntary listing pathway for medical devices and IVDs. This article outlines the regulatory landscape, classification, the role of the Local Responsible Person (LRP), and the registration process for market entry, all based on our insights and experience for efficient Hong Kong medical device registration.
Read article →
June 23, 2025
Medical Device and IVD Registration & Approval in Hong Kong
Hong Kong's Medical Device Administrative Control System (MDACS) offers a voluntary listing pathway for medical devices and IVDs, with a future mandatory framework on the horizon. This article explores the classification system, the crucial role of the Local Responsible Person (LRP), and the registration process for market entry, all based on our insights and experience for efficient Hong Kong medical device and IVD registration.
Read article →
June 23, 2025
Code of Practice for Listed Distributors of Medical Devices in Hong Kong: A Guide to COP-05
For medical device distributors listed under Hong Kong's MDACS, adhering to a specific Code of Practice is crucial for maintaining effective distribution channels and public safety. This article, guided by COP-05, details their responsibilities in areas such as record keeping, handling, and post-market vigilance, all from our experience and insights.
Read article →
June 23, 2025
Code of Practice for Listed Importers of Medical Devices in Hong Kong: A Guide to COP-04
For medical device importers listed under Hong Kong's MDACS, adhering to a specific Code of Practice is vital for ensuring supply chain integrity and public safety. This article, guided by COP-04, details their responsibilities in areas such as record keeping, handling, and post-market vigilance, all from our experience and insights.
Read article →
June 23, 2025
Code of Practice for Listed Local Manufacturers of Medical Devices in Hong Kong: A Guide to COP-03
Local manufacturers of medical devices listed under Hong Kong's MDACS adhere to a Code of Practice ensuring quality and safety. This article, guided by COP-03, outlines their responsibilities, focusing on quality management systems, production control, and post-market vigilance to uphold regulatory standards, all from our experience and insights.
Read article →
June 23, 2025
Code of Practice for Conformity Assessment Bodies (CABs) in Hong Kong: A Guide to COP-02
Conformity Assessment Bodies (CABs) play a vital role in verifying medical device compliance in Hong Kong's MDACS. This article, guided by Code of Practice COP-02, outlines the recognition process, responsibilities, and operational requirements for CABs to ensure objective and rigorous assessments of medical device safety and performance, all from our experience and insights.
Read article →
June 23, 2025
Code of Practice for Local Responsible Persons (LRPs) in Hong Kong: A Guide to COP-01
The Local Responsible Person (LRP) is a cornerstone of Hong Kong's Medical Device Administrative Control System (MDACS). This article, guided by Code of Practice COP-01, outlines the extensive responsibilities and duties of LRPs in ensuring the safety, quality, and performance of medical devices throughout their lifecycle, all from our experience and knowledge.
Read article →
June 23, 2025
Personalised Medical Devices in Hong Kong: A Guide to TR-009
As medical technology advances towards individualized patient care, Hong Kong's MDACS has issued specific guidance on Personalised Medical Devices. This article, guided by Technical Reference TR-009, defines and categorizes these devices—Custom-Made, Patient-Matched, and Adaptable—and clarifies their regulatory applicability, all from our experience and insights.
Read article →
June 23, 2025
Artificial Intelligence Medical Devices (AI-MD) in Hong Kong: A Guide to TR-008
As AI transforms healthcare, Hong Kong's MDACS provides specific guidance for Artificial Intelligence Medical Devices (AI-MDs). This article, guided by Technical Reference TR-008, covers the definitions, unique regulatory considerations, and essential documentation requirements for AI-MDs, including those with Continuous Learning Capability (CLC), all from our experience and insights.
Read article →

June 23, 2025
Essential Principles of Safety and Performance for Medical Devices in Hong Kong: A Deep Dive into TR-004
The foundation of medical device regulation in Hong Kong's MDACS is built upon the Essential Principles of Safety and Performance. This article, specifically guided by Technical Reference TR-004, details these fundamental requirements that manufacturers must integrate into every stage of a device's lifecycle to ensure patient and user safety, all based on our insights and experience for robust medical device regulation in Hong Kong.
Read article →
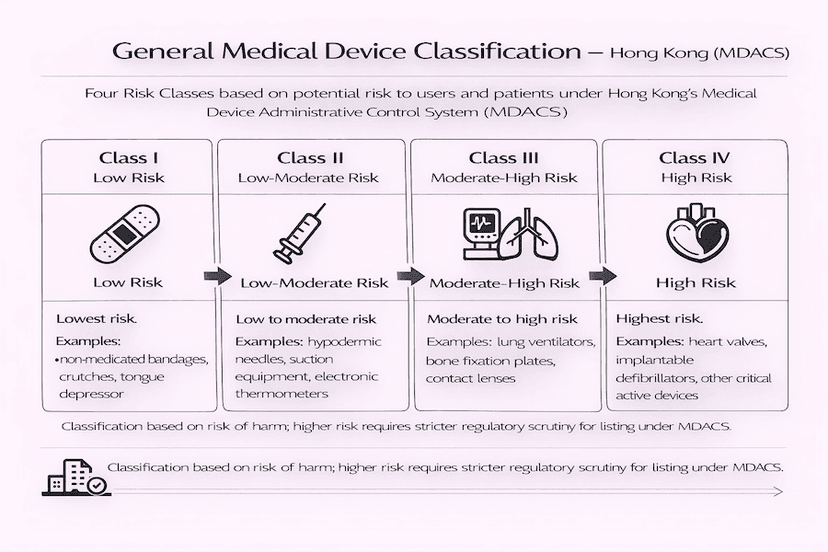
June 23, 2025
Classification of General Medical Devices in Hong Kong: A Guide to TR-003
Understanding the risk-based classification of general medical devices is fundamental for market access in Hong Kong. This article, guided by Technical Reference TR-003, details the rules and factors that determine a device's risk class under the Medical Device Administrative Control System (MDACS), all based on our insights and experience for efficient medical device classification in Hong Kong.
Read article →

June 23, 2025
Summary Technical Documentation (STED) for Medical Devices in Hong Kong: A Guide to TR-002
The Summary Technical Documentation (STED) is a crucial component of medical device listing applications under Hong Kong's MDACS. This article, guided by Technical Reference TR-002, explains the purpose and required content of STED for demonstrating conformity to Essential Principles of Safety and Performance, all based on our insights and experience for streamlined medical device documentation in Hong Kong.
Read article →
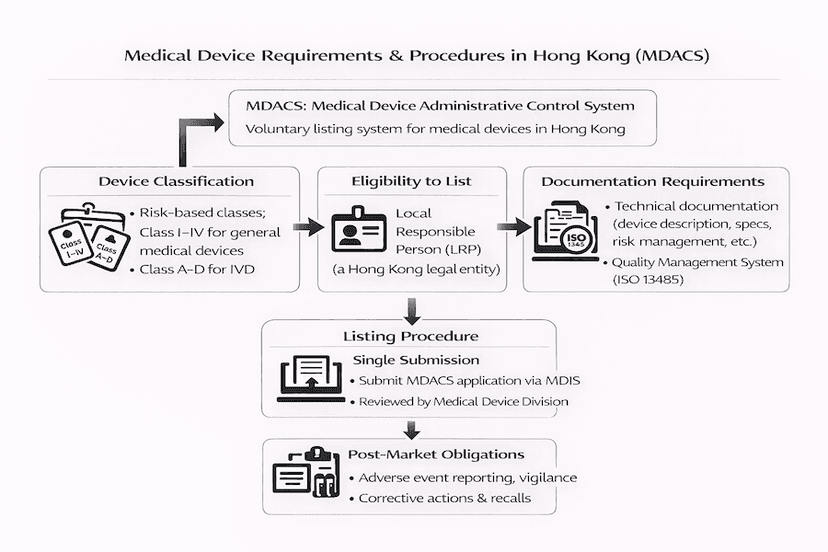
June 23, 2025
Medical Device Registration in Hong Kong: Essential Requirements and Procedures
A comprehensive guide to the essential requirements and procedures for medical device registration under Hong Kong's Medical Device Administrative Control System (MDACS), all based on our insights and experience for efficient Hong Kong medical device registration.
Read article →
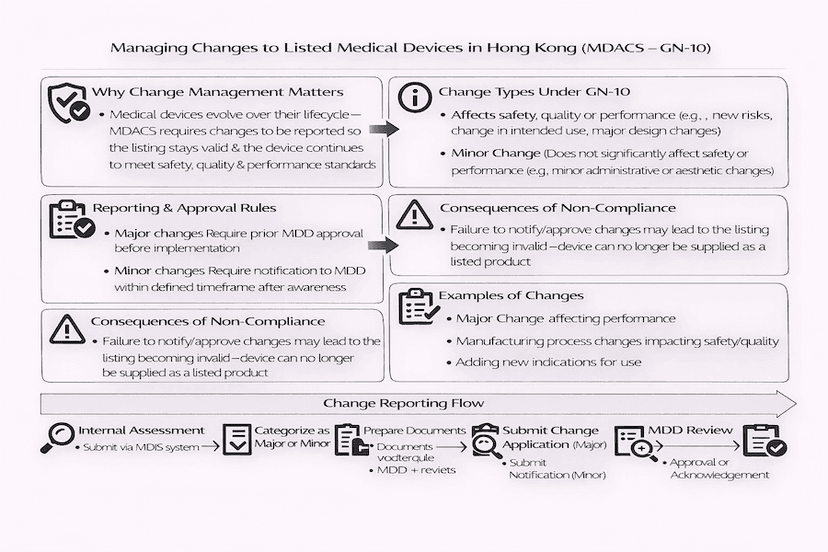
June 16, 2025
Managing Changes for Listed Medical Devices in Hong Kong: A Guide to GN-10
Changes to listed medical devices under Hong Kong's Medical Device Administrative Control System (MDACS) require careful management and reporting. This article, guided by GN-10, explains how Local Responsible Persons (LRPs) should categorize, manage, and report major and minor changes to ensure continued compliance and patient safety, based on our insights.
Read article →
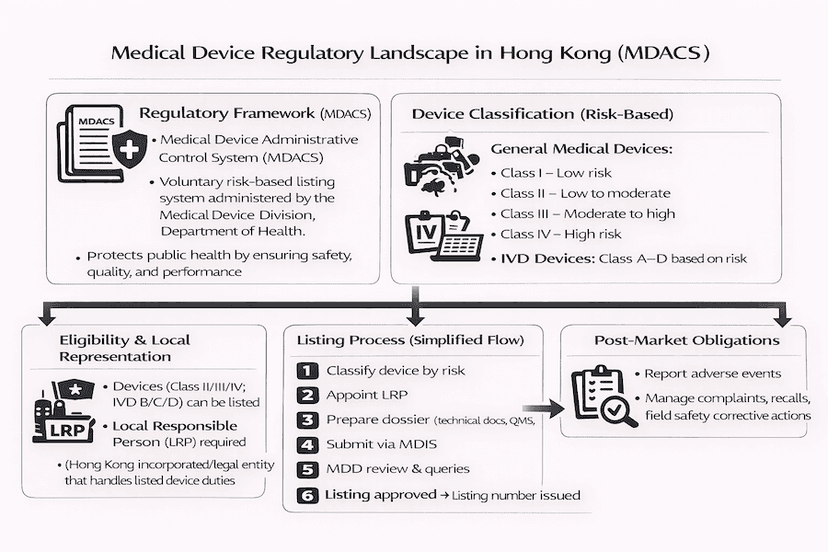
June 16, 2025
Navigating the Medical Device Regulatory Landscape in Hong Kong
A detailed overview of the Medical Device Administrative Control System (MDACS) in Hong Kong, covering device classification, the comprehensive listing process, post-market obligations, and the future of mandatory regulation, shared from our insights and experience.
Read article →
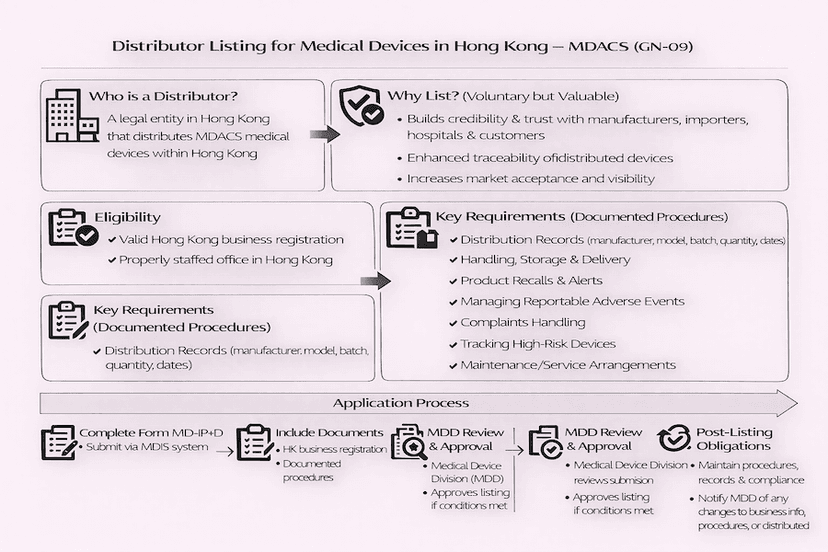
June 15, 2025
Listing Procedures for Medical Device Distributors in Hong Kong: A Guide to GN-09
This article outlines the application process for medical device distributors seeking to be listed under Hong Kong's Medical Device Administrative Control System (MDACS), as detailed in GN-09. It covers eligibility, key requirements for documented procedures, and the submission process to enhance traceability and public safety, based on our insights.
Read article →
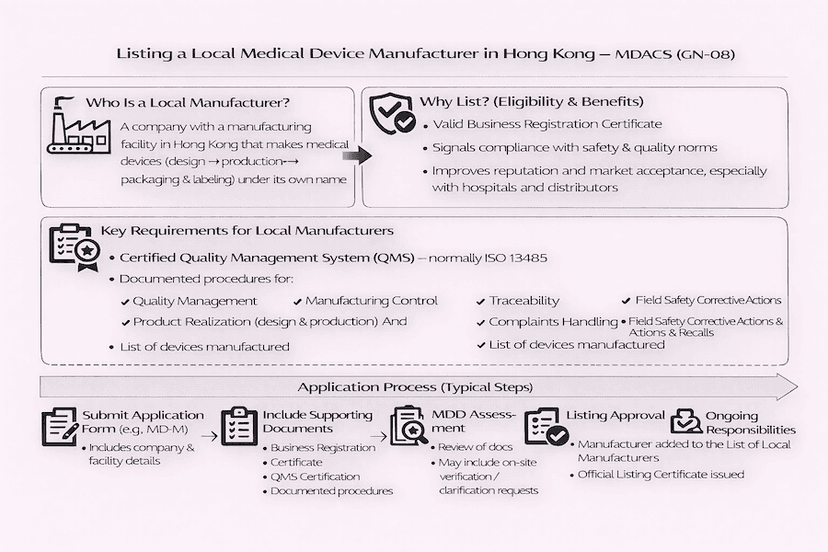
June 14, 2025
Listing Procedures for Local Medical Device Manufacturers in Hong Kong: A Guide to GN-08
This article details the application process for local medical device manufacturers seeking to be listed under Hong Kong's Medical Device Administrative Control System (MDACS), as guided by GN-08. It covers eligibility, quality management system requirements, and the submission process, all based on our insights and experience for effective Hong Kong medical device manufacturing.
Read article →
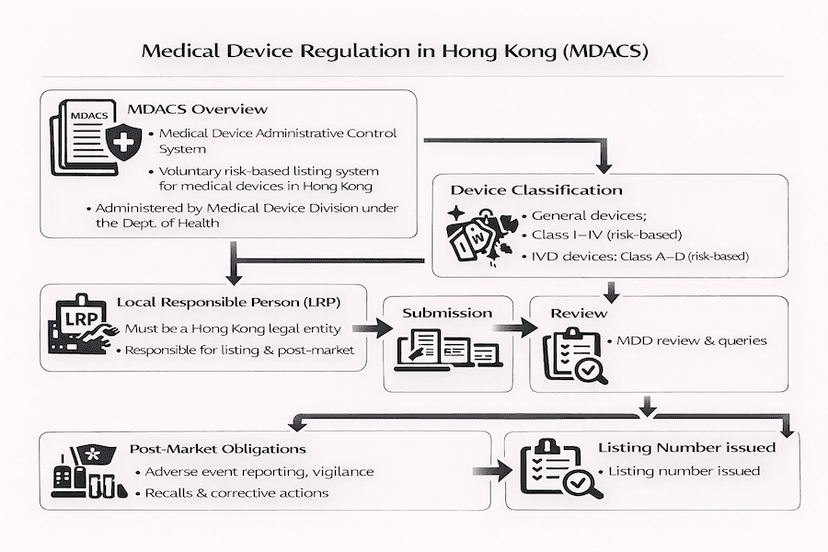
June 14, 2025
Navigating Medical Device Regulations in Hong Kong: The MDACS Framework
Hong Kong's Medical Device Administrative Control System (MDACS) provides a robust, although currently voluntary, framework for regulating medical devices. This article explores the system's key features, including device classification, the listing process, the crucial role of Local Responsible Persons (LRPs), and its increasing importance for market access and public procurement, all from our insights and experience.
Read article →
June 2, 2025
Listing Procedures for Class B, C, and D In Vitro Diagnostic Medical Devices in Hong Kong: A Guide to GN-06
This article details the application process for listing Class B, C, and D In Vitro Diagnostic Medical Devices (IVDMDs) under Hong Kong's Medical Device Administrative Control System (MDACS), as guided by GN-06. It covers classification, eligibility, submission requirements, and the online application via MDIS, all based on our insights and experience for efficient IVDMD listing in Hong Kong.
Read article →
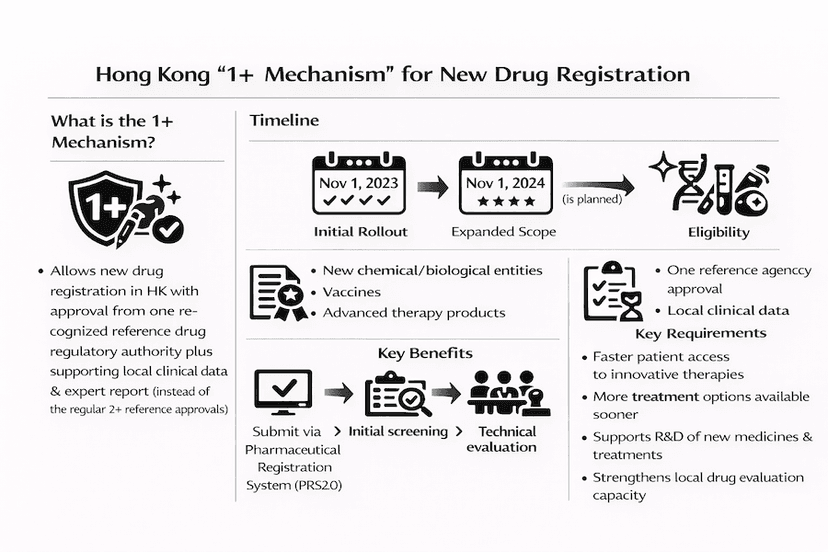
June 1, 2025
The "1+" Mechanism: Accelerating New Drug Registration in Hong Kong
Learn about Hong Kong's innovative "1+" mechanism for drug registration, its impact on patient access to new treatments, and its role in promoting healthcare development in the Greater Bay Area, all through our insights and experience.
Read article →

May 24, 2025
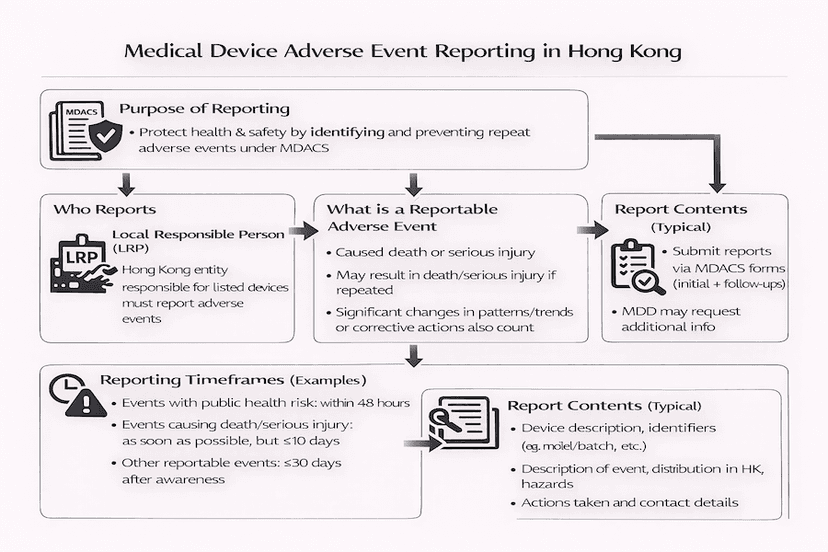
Medical Device Adverse Event Reporting in Hong Kong: A Guide for LRPs
Adverse event reporting is a critical component of Hong Kong's Medical Device Administrative Control System (MDACS), aiming to enhance public health and safety. This article outlines the requirements and responsibilities of Local Responsible Persons (LRPs) in reporting adverse events related to listed medical devices, based on our insights.
Read article →
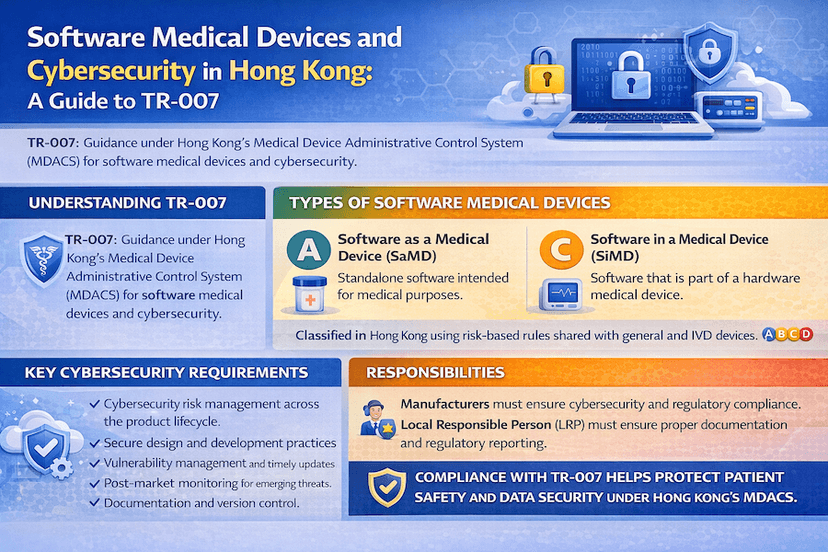
May 17, 2025
Software Medical Devices and Cybersecurity in Hong Kong: A Guide to TR-007
As software plays an increasingly vital role in healthcare, Hong Kong's MDACS addresses its regulation, including cybersecurity. This article, guided by Technical Reference TR-007, outlines the definition of Software as a Medical Device (SaMD) and Software in a Medical Device (SiMD), and the crucial cybersecurity considerations for manufacturers and LRPs, all based on our insights and experience for robust Hong Kong medical device cybersecurity.
Read article →
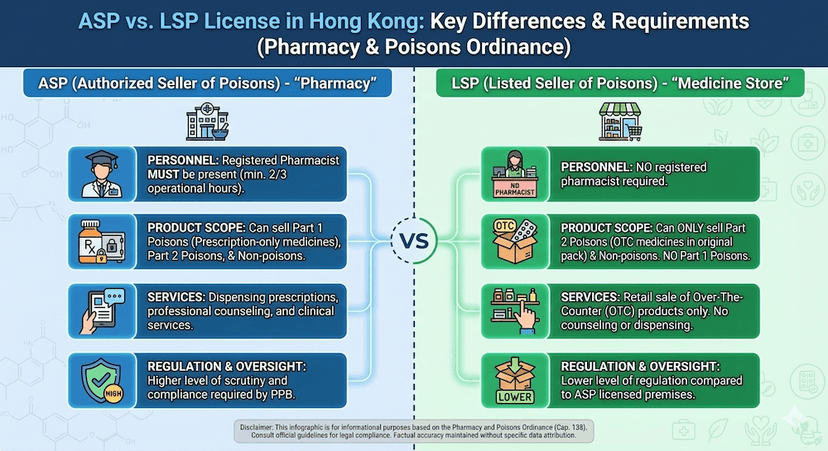
May 10, 2025
ASP vs LSP License: Which One Do You Need?
Learn the differences between Authorized Seller of Poisons (ASP) and Listed Seller of Poisons (LSP) licenses and determine which is right for your business.
Read article →
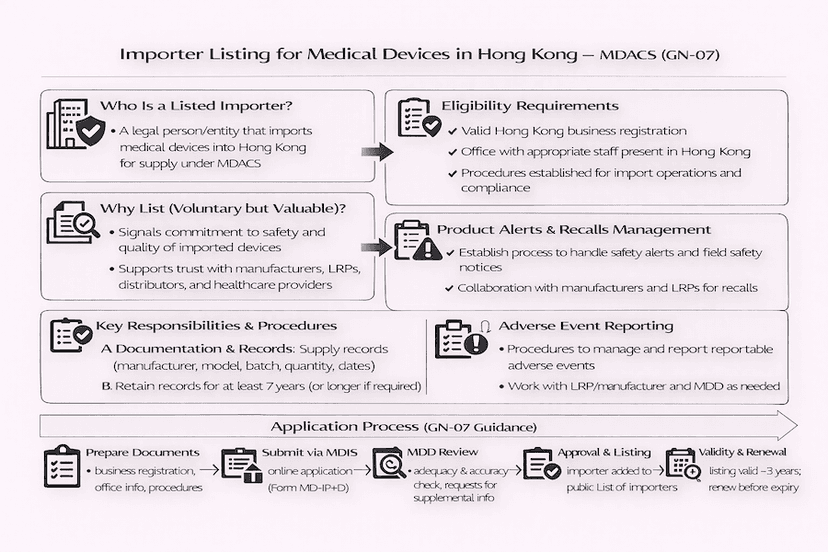
May 3, 2025
Listing Procedures for Medical Device Importers in Hong Kong: A Guide to GN-07
For entities importing medical devices into Hong Kong, the Medical Device Administrative Control System (MDACS) provides a voluntary listing scheme for importers, guided by GN-07. This article details the eligibility, application steps, and key requirements for listing as a medical device importer, based on our insights and experience for efficient Hong Kong medical device import.
Read article →
April 18, 2025
Medical Device Registration in Hong Kong: A Market and Regulatory Overview
Hong Kong presents a growing and dynamic market for medical devices, governed by the Medical Device Administrative Control System (MDACS). This article outlines the regulatory landscape, key market trends, device classification, and the general process for registration and market access in this evolving region, all based on our insights and experience.
Read article →
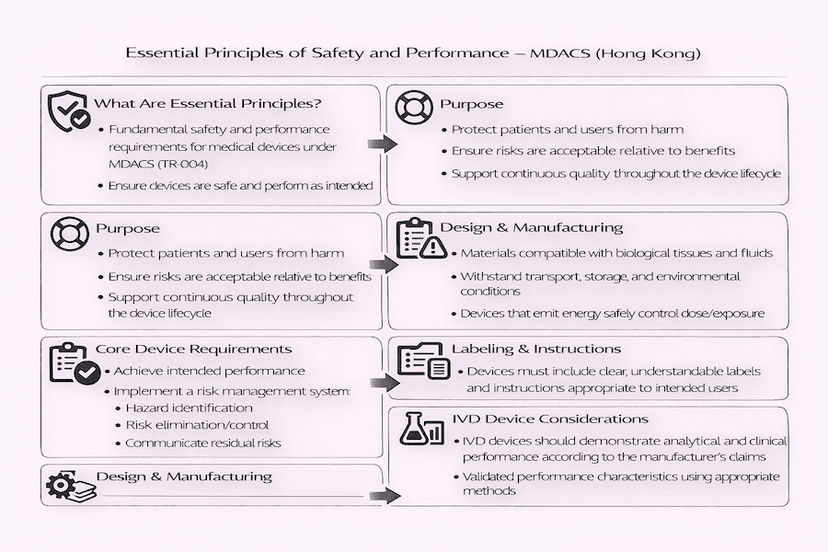
April 17, 2025
Essential Principles of Safety and Performance for Medical Devices in Hong Kong: Understanding TR-001 and TR-004
The safety and performance of medical devices in Hong Kong are underpinned by a set of 'Essential Principles,' detailed in Technical References TR-001 and TR-004 of the MDACS. This article explores these fundamental requirements that manufacturers must meet in the design, manufacture, and post-market phases of medical devices, from our insights and experience for robust medical device regulation in Hong Kong.
Read article →
April 12, 2025
Medical Device Product Registration in Hong Kong: A Focused Approach
Navigating medical device regulatory compliance in Hong Kong is crucial for market access. This article highlights services focused on preparing products for the Medical Device Administrative Control System (MDACS) and streamlining the registration process for efficient market entry, based on our insights.
Read article →
April 6, 2025
Global Medical Device Registration: Regulatory Expectations Across Key Markets
Bringing a single medical device to multiple markets requires understanding varied regulatory expectations, which are far from a "copy and paste" exercise. Key differences include the EU's focus on clinical evidence (MDR), the USA's reliance on substantial equivalence (510(k)), and Japan's highly process-driven review, emphasizing the need for robust global strategy and potential leveraging of programs like **MDSAP**.
Read article →
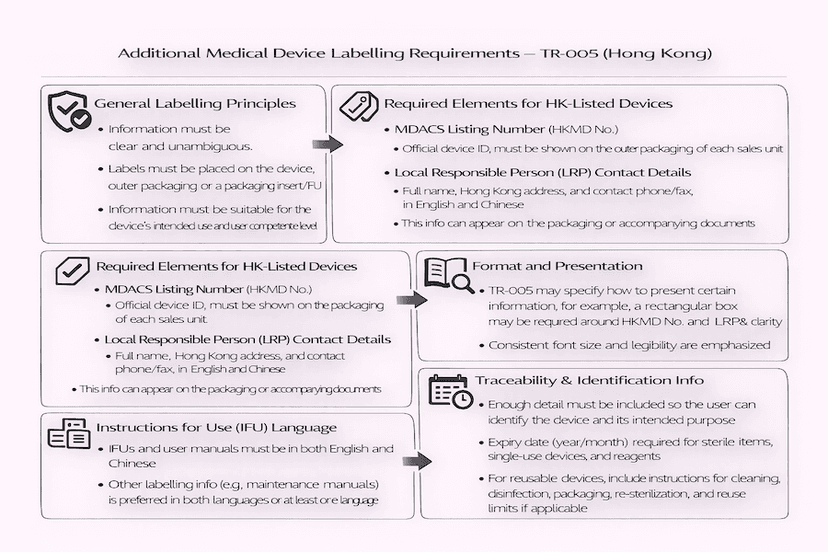
March 21, 2025
Additional Medical Device Labelling Requirements in Hong Kong: Understanding TR-005
Beyond the general Essential Principles, Hong Kong's Medical Device Administrative Control System (MDACS) imposes specific additional labelling requirements for medical devices. This article, guided by Technical Reference TR-005, details these crucial elements to ensure clear communication and compliance in the local market, all based on our insights and experience for effective medical device labelling in Hong Kong.
Read article →
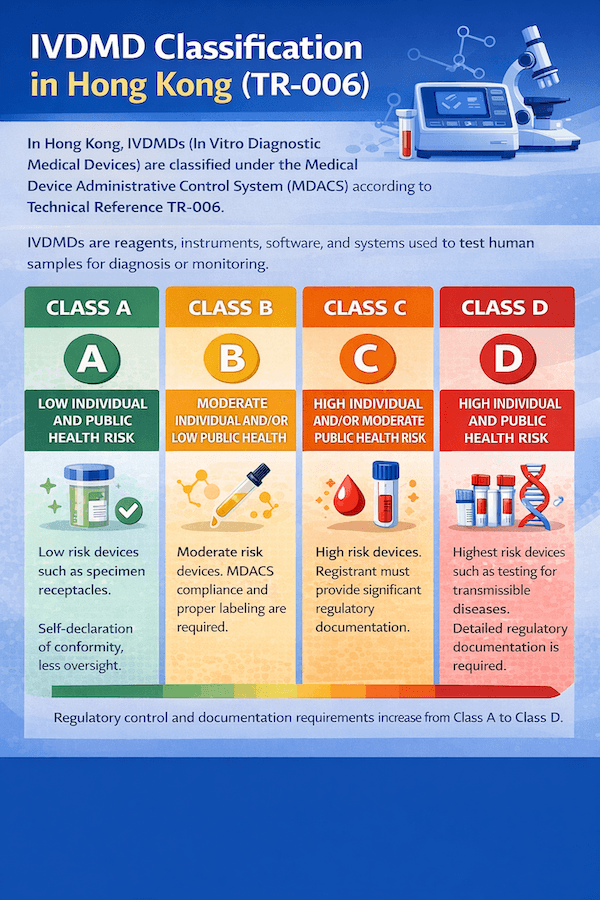
March 17, 2025
Classification of In Vitro Diagnostic Medical Devices (IVDMDs) in Hong Kong: A Guide to TR-006
The classification of In Vitro Diagnostic Medical Devices (IVDMDs) in Hong Kong is a risk-based system crucial for regulatory compliance. This article, guided by Technical Reference TR-006, outlines the principles and categories that determine an IVDMD's risk class under the Medical Device Administrative Control System (MDACS), all based on our insights and experience for efficient IVDMD classification in Hong Kong.
Read article →
March 5, 2025
Digital Transformation in Pharmacy Management
How modern software solutions are revolutionizing pharmacy operations, improving efficiency, and enhancing patient care in the digital age.
Read article →

January 28, 2025
Pharmacovigilance Best Practices in Hong Kong
Essential guidelines for adverse event reporting and pharmacovigilance compliance in Hong Kong's pharmaceutical industry.
Read article →
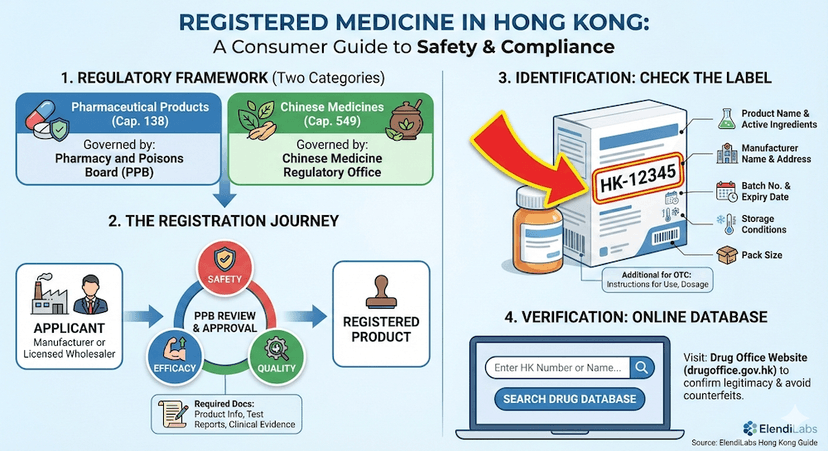
January 16, 2025
Understanding Registered Medicine in Hong Kong: A Complete Guide
Learn about Hong Kong's medicine registration system, legal requirements, and how to identify registered medicines to ensure safety and efficacy.
Read article →
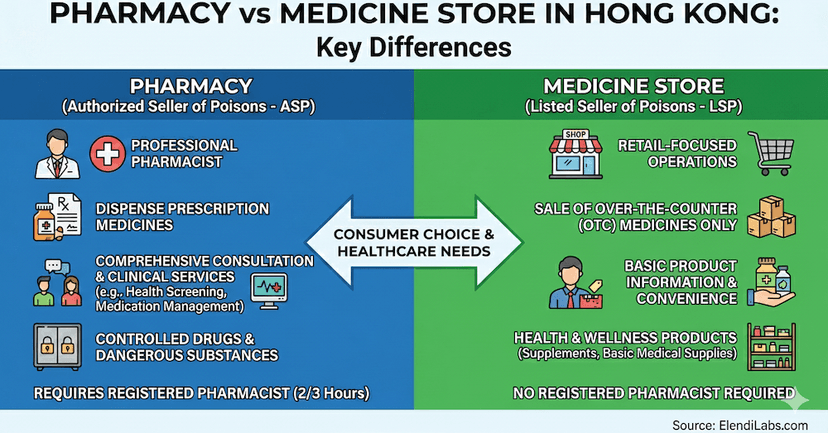
January 15, 2025
Pharmacy vs Medicine Store: Understanding the Key Differences
A pharmacy has a licensed pharmacist to dispense prescription drugs, while a medical store sells OTC items. Discover key legal and service differences here.
Read article →

December 20, 2024
Import and Export Regulations for Pharmaceuticals
Navigate the complex landscape of pharmaceutical import and export regulations in Hong Kong with this detailed guide.
Read article →
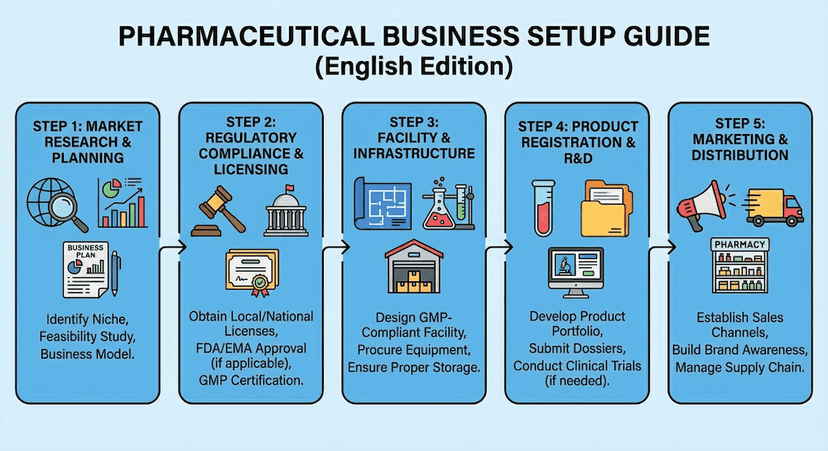
October 15, 2024
Setting Up Your Pharmaceutical Business in Hong Kong
Step-by-step guide to establishing a pharmaceutical business in Hong Kong, from licensing to operational requirements.
Read article →
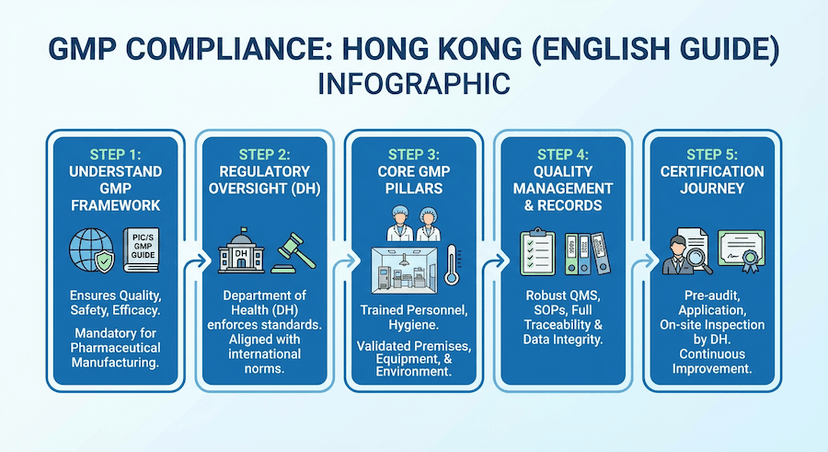
August 10, 2024
Good Manufacturing Practice (GMP) Compliance in Hong Kong
Essential requirements and best practices for implementing GMP compliance in pharmaceutical manufacturing facilities.
Read article →
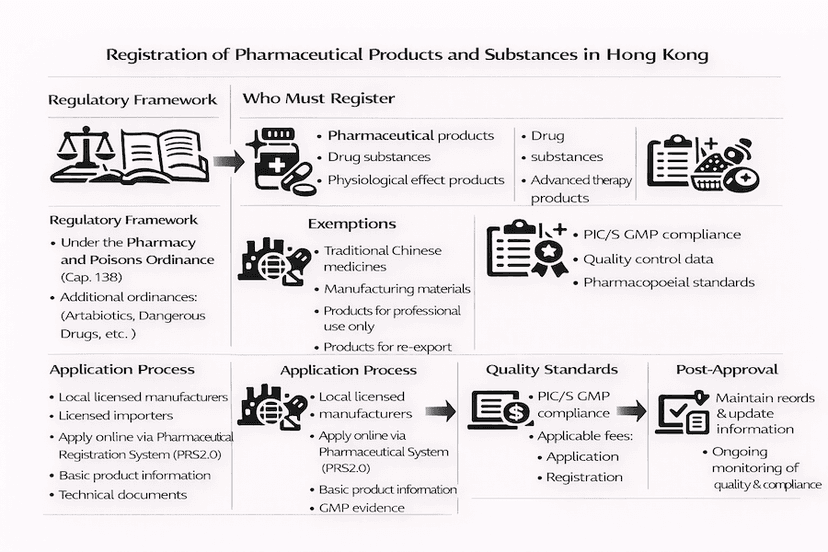
July 20, 2024
Professional Guide: Registration of Pharmaceutical Products and Substances in Hong Kong
A comprehensive guide for healthcare professionals and industry stakeholders on the registration requirements, procedures, and compliance for pharmaceutical products and substances in Hong Kong.
Read article →
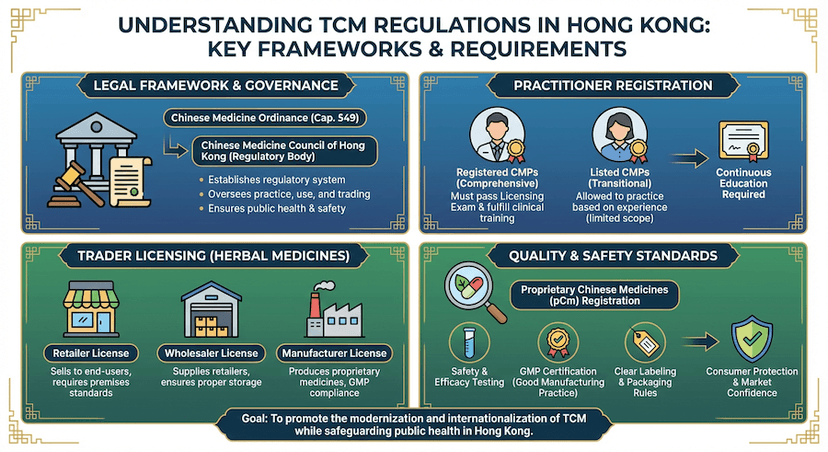
June 5, 2024
Traditional Chinese Medicine (TCM) Regulations in Hong Kong
Comprehensive guide to Traditional Chinese Medicine regulations, including practitioner registration and Chinese medicine licensing.
Read article →
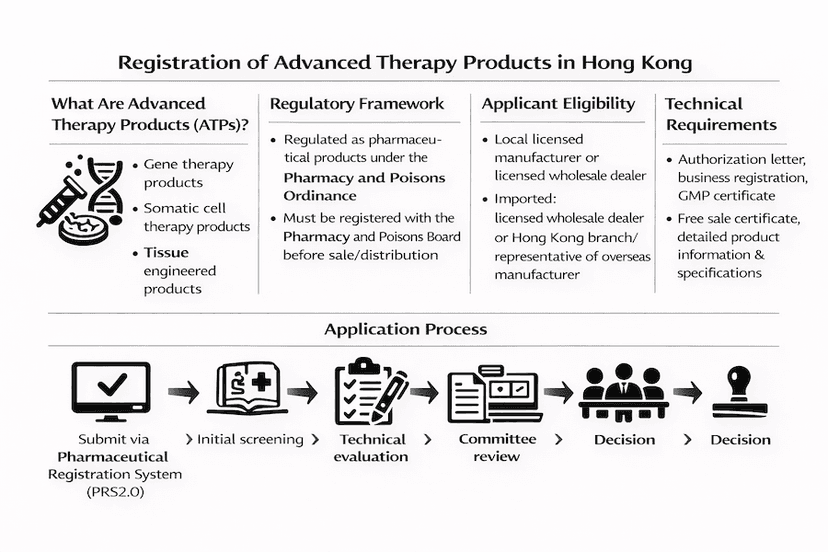
May 10, 2024
Registration of Advanced Therapy Products in Hong Kong: A Comprehensive Guide
Learn about the registration process for advanced therapy products (ATPs) in Hong Kong, including gene therapy, somatic cell therapy, and tissue engineered products.
Read article →
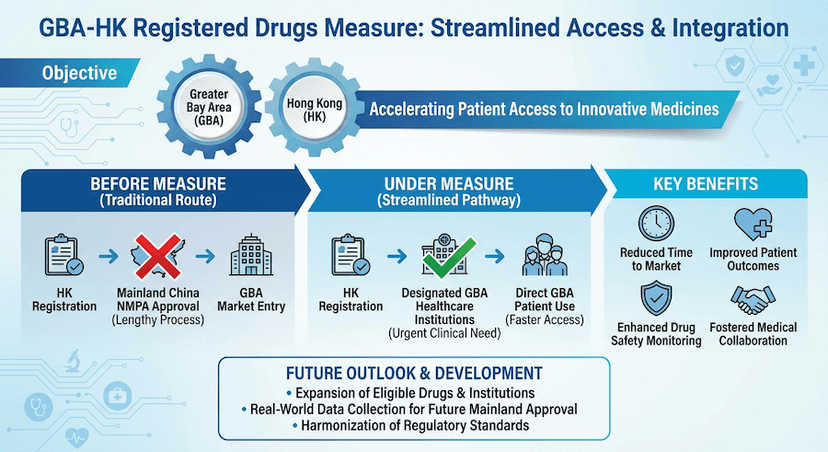
March 20, 2024
Using Hong Kong Registered Drugs and Medical Devices in the Greater Bay Area: A Comprehensive Guide
Learn about the innovative measure that allows designated healthcare institutions in the Greater Bay Area to use Hong Kong-registered drugs and medical devices, facilitating better healthcare access for Hong Kong residents.
Read article →
March 20, 2024
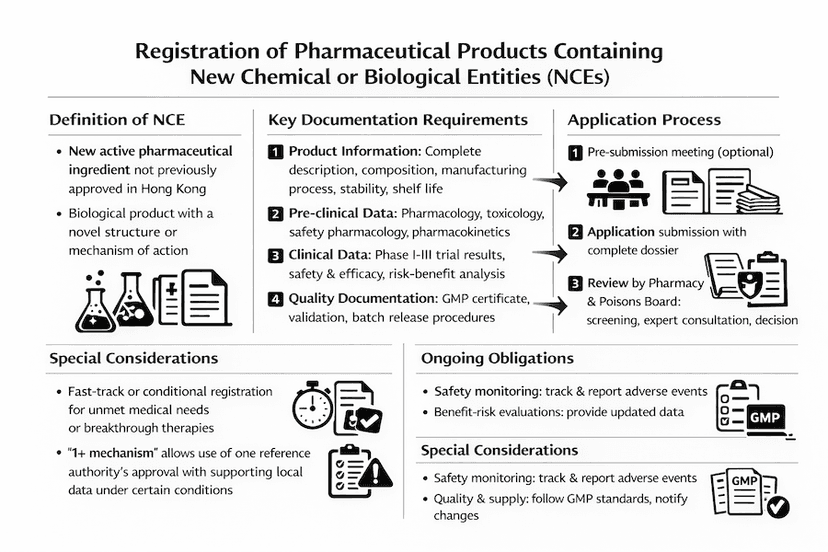
Registration of Pharmaceutical Products Containing a New Chemical or Biological Entity in Hong Kong
A comprehensive guide to the registration process for pharmaceutical products containing new chemical or biological entities in Hong Kong, including requirements, procedures, and regulatory considerations.
Read article →
February 20, 2024
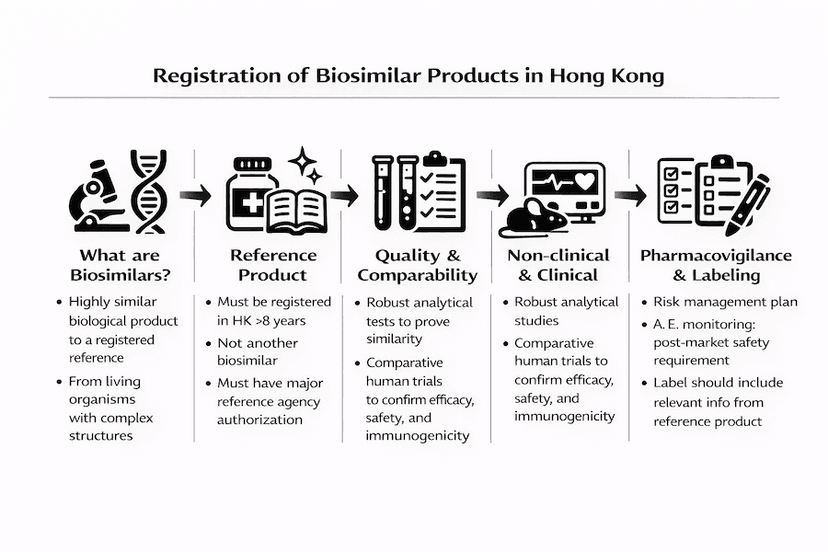
Registration of Biosimilar Products in Hong Kong: A Comprehensive Guide
Learn about the registration process for biosimilar products in Hong Kong, including requirements, documentation, and special considerations for these complex biological medicines.
Read article →